Clinical Trial:
Lactobacillus fermentum ME-3
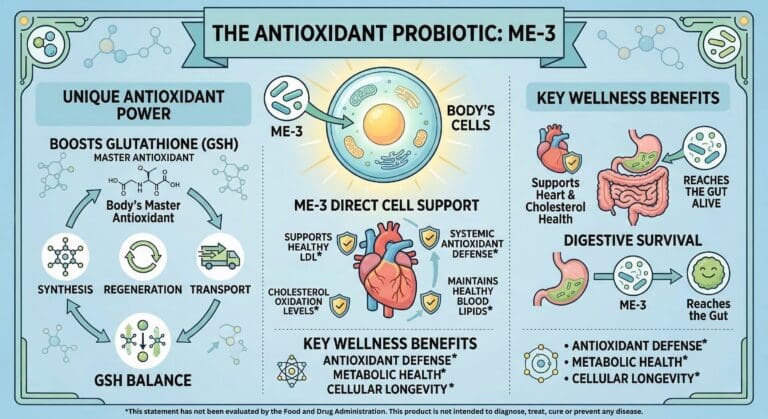
Lactobacillus fermentum ME-3 (often referred to as the “antioxidant probiotic“) is one of the most clinically researched probiotic strains, particularly regarding its unique ability to stimulate the body’s internal antioxidant systems.
Discovered in 1995 at the University of Tartu, Estonia, it is the only known probiotic that functions as a “complete glutathione system”—it can synthesize, take up, and recycle glutathione, the body’s master antioxidant.
Key Human Clinical Trial Areas
- Cardiovascular & Lipid Profile
The most extensive clinical evidence for ME-3 is in cardiovascular health. Multiple double-blind, placebo-controlled trials have demonstrated its impact on lipid markers:
- Cholesterol Reduction: Trials (e.g., ISRCTN49744186) showed a 12% to 17% reduction in LDL cholesterol after 4–8 weeks of supplementation.
- Oxidized LDL (oxLDL): A significant marker for atherosclerosis. Studies showed a 16% reduction in oxLDL.
- Triglycerides: Reductions of approximately 10% to 12% were observed in subjects with borderline-high levels.
- Paraoxonase (PON1): Clinical intake led to an 18% increase in PON1 activity, an enzyme that protects HDL (“good”) cholesterol from oxidation.
- Metabolic Health & Diabetes
Recent clinical reviews (up to 2025/2026) highlight ME-3’s role in metabolic syndrome parameters:
- HbA1c Reduction: In a 4-week trial, subjects saw a significant decrease in glycated hemoglobin (HbA1c), suggesting better long-term blood sugar regulation.
- Inflammation: Clinical trials reported substantial drops in inflammatory markers: 31% reduction in hs-CRP and 22% reduction in Interleukin-6 (IL-6).
- Body Composition: Some 2026 reports (e.g., regarding the PrimeBiome formulation) indicate that ME-3 may assist in reducing visceral fat mass by modulating gut-derived hormones related to satiety.
- Antioxidant Status & Detoxification
Clinical trials have confirmed that ME-3 directly improves the Glutathione (GSH) Ratio:
- GSH/GSSG Ratio: Studies showed a 49% increase in the ratio of reduced to oxidized glutathione, indicating a much higher capacity for the body to fight oxidative stress.
- Detoxification: By boosting glutathione, trials suggest ME-3 supports liver function, particularly in neutralizing heavy metals (like mercury and lead) and environmental toxins.
- Stroke Recovery & Neurological Health
A specialized clinical trial involving elderly patients recovering from ischemic stroke found that ME-3 supplementation:
- Significantly improved scores on the Scandinavian Stroke Scale.
- Improved the Functional Independence Measure (FIM).
- Reduced markers of post-stroke oxidative damage in the blood.
Trial Summaries & Registries
| Study/ID | Focus Area | Duration | Key Result |
|---|---|---|---|
| ISRCTN49744186 | Borderline-High Lipids | 8 Weeks | ↓Triglycerides (17%), ↓LDL (5%) |
| ISRCTN55339917 | Reg’Activ Cholesterol | 4 Weeks | ↓Total Cholesterol (12%), ↓hs-CRP (31%) |
| Kullisaar et al. | Post-prandial Oxidative Stress | 2 Weeks | ↓Post-meal Oxidative Stress & Triglyceride spike |
| Tartu University | Stroke Recovery | 3 Weeks | Improved cognitive & physical functional recovery |
Safety Profile
In all clinical trials to date, L. fermentum ME-3 has been shown to be well-tolerated with no significant adverse effects. It holds QPS (Qualified Presumption of Safety) status in Europe and is widely used in commercial formulations like Reg’Activ and PrimeBiome.

